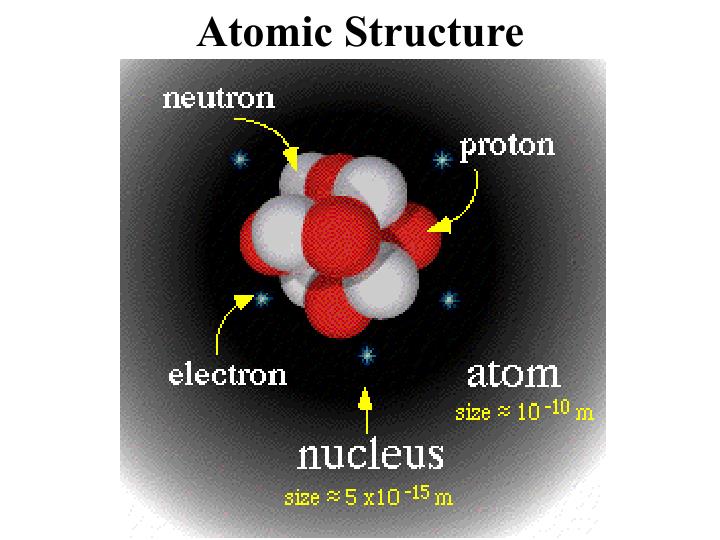 
most iron nuclei with 26 protons contain 30 neutrons, while a small percentage contain 29 neutrons.small percentage contain one neutron and a smaller percentage, two neutrons.all hydrogen nuclei have a single proton but most have no neutrons.number of protons in the nucleus need not match the number of neutrons there.number of protons in a nucleus exactly matches the number of electrons around the nucleus in a neutral atom,.mass of an electron is so small that it's disregarded.mass of an atom in atomic mass units is simply the sum of its protons and neutrons and is known as the atomic mass number.approximate mass of a single proton or neutron is 1 amu.(Nucleus) BB on the 50 yard line with the electrons at a distance of about 50 yards from the BB.Doritos are orbiting the cars at a distance of a few miles.Electrons are like bags of doritos (1 lb objects).Nucleons (protons and neutrons) are like toyotas (2000 lb cars).About 6 years for one of your exhaled breaths to become mixed in the atmosphere As many atoms in a breath of air as breathfuls of air in Earth’s atmosphere.About 1023 atoms in a thimbleful of water.Strong nuclear force – only apparent at distances of 1 – 2 femtometers (10-15m).Since like charges repel, how are the protons bound together in the nucleus?.Actually, protons and neutrons can be further split into quarks.Atoms composed of protons, neutrons, and electrons.In particle accelerators and in stars, subatomic particles can be found.Greek origin – atomos which means indivisible.Smallest particle of an element that maintains the properties of the element.Taken with a scanning tunneling microscope at the IBM Almaden Laboratory in San Jose, CA.Iron atoms positioned into a circular ring that “corrals” electrons.Strings of dots are chains of thorium atoms.Photo made in 1970 with a scanning electron microscope by researchers at the University of Chicago's Enrico Fermi Institute.Hydrogen is most abundant element in universe.Life – mostly oxygen, carbon, hydrogen, nitrogen, & calcium.Greater than 99% of matter on earth is formed by about a dozen elements.The remaining elements are made in laboratories and are too unstable (radioactive) to occur in nature in significant amounts.Only about 118 different elements make up all the materials we know.The structure of the universe includes the structure of the atom, and of the particles making up the atom, and the properties of the atom, and of the particles making up the atom.- E N D - Presentation Transcript The order of discover is inevitable and set by the structure of the universe. The charges of particles, their mass and ability to survive outside the particles they make up, and other properties will make a particle harder or easier to discover. The order of discovery of particles is also affected by the properties of the particles. 
The sub atomic particles were discovered in a necessary order with the outer particles like the electron being discovered earlier, and inner particles such as quarks being discovered later. This led to the idea there was a separate atom for each element which explained the differences between the elements. The traditional view of the elements could be obtained by naked eye observation, and the view of nature as being made up of the chemical elements in the periodic table was next discovered, as it involved the decomposition of traditional elements, such as air and water. This was because nature has a particular structure and we have a particular place in nature. The order of discovery of these ideas of the ultimate constituents of matter was necessary, in that they could not have been discovered in any other order. The change from the traditional Western and Chinese view of the elements involving materials such as water, air, earth, wood, metal and fire, to the chemical elements making up the periodic table, to atoms, to particles such as protons, neutrons and electrons, and then to quarks was inevitable.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
